News
Underlying mechanisms in latent infections
Dov B. Prusky et al. analyze if quiescence of postharvest pathogens is a fungal inhibition process or an immune response of the unripe host fruit
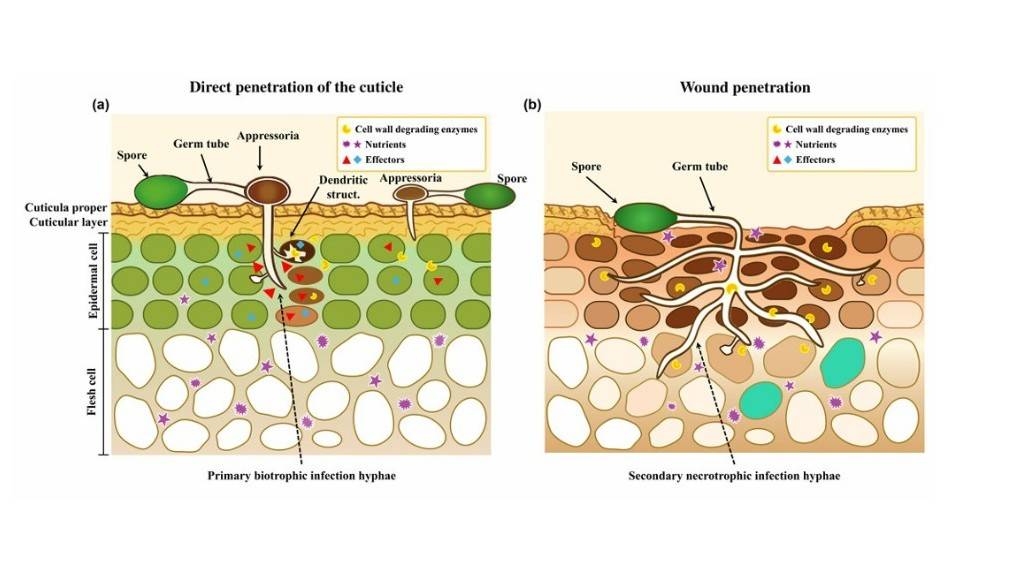
Postharvest pathogens can infect fresh produce both before and after harvest, by direct or wound-enhanced penetration, remaining quiescent until ripening.
Biotrophic-like postharvest pathogens persist beneath host cells and can remain in a state of quiescence. They detect environmental cues and regulate quiescence through chromatin-level control and the secretion of effectors that interact with host pattern recognition receptors.
Bycontrast, necrotrophic fungi persist between dead cells and depend more directly on nutrient availability to prime their growth and upon secretion fo rfungal virulence factors.
During quiescence, the host also mounts specific responses, including
- activation of pattern recognition receptor genes,
- ethylene signaling (particularly in unripe fruit), and
- defense genes such as PR-10 and chitinases.
Jasmonic acid and ethylene pathways synergistically enhance these defenses.
As fruit ripens, the transition from quiescence to active necrotrophic growth istriggered, accelerating tissue decay.
This activation is driven by several key factors, including
- weakened host defenses,
- decreased levels of antifungal compounds such as polyphenols,
- increased cell wall accessibility due to fruit softening and
- ripening-associated changes in signaling pathways, which alter environmental pH, carbon metabolism, and secondary metabolite production.
These regulatory mechanisms collectively govern the timing and extent of fungal initiation of colonization during fruit senescence.
Introduction
Fruits and vegetables are highly perishable commodities, and improper handling during harvesting, transportation, or storage can result in significant postharvest losses and waste.
The Food and Agriculture Organization (FAO) of the United Nations
estimates that about one-third of global food production– c. 1.3 billion metric tons– is lost or wasted each year (Sagar et al., 2018).
In the case of horticultural products, losses can reach up to 60%, occurring at various stages of the supply chain, from harvest to household consumption (Prusky & Roma nazzi, 2023).
Postharvest fungal infections may be initiated before, during, or after harvest but often remain dormant in a quiescent stage until fruit ripening and senescence (Prusky, 1996; Prusky et al., 2013).
This is different from fungal pathogens that show a complete full cycle in living tissue; for example, Ustilago (Yu et al., 2023), which are not covered here.
Disease prevention is typically achievedthrough a combination of fungicide treatments and optimized storage conditions (Adaskaveg et al., 2023).
Symptoms usually become visible only after prolonged cold storage or during shel flife, just before consumption.
Notably, while most freshly harvested fruits and vegetables harbor quiescent infections, the mechanisms governing fungal quiescence and host resistance in unripe fruits remain poorly understood (Adaskaveg et al., 2000; Prusky et al., 2013).
Although recent advances have introduced stable and robust sensing materials with high sensitivity for detecting fruit infections by pathogens, no practical applications have been implemented to date (Archana et al., 2024).
This knowledge gap underscores the importance of investigating the factors that regulate quiescence and its activation, as such insights could inform the development of novel disease management strategies aimed at reducing reliance on postharvest fungicides while preserving fruit quality over time (Prusky et al., 2013).
What are the infection mechanisms of postharvest pathogens that become quiescent
Postharvest pathogens infect fruits and vegetables through two main mechanisms:
- direct penetration of the cuticle or
- entry through wounds,
both of which can occur at pre- and postharvest stages.
Fungal host specificity refers to a pathogen’s ability to infect only particular host species, ranging from narrow specificity– such as Penicillium digitatum infecting only citrus fruit– to broad specificity, as seen with Botrytis cinerea, which infects a wide range of fruit and vegetable hosts (Adaskaveg et al., 2023).
When a host specific postharvest pathogen attempts to infect a nonhost species, the expected outcome is pathogen death and effective host immunity.
Host specificity is governed by a complex interplay of genetic, structural, and environmental factors, including the pathogen’s capacity to overcome host defenses and the host’s inherent genetic resistance mechanisms (Jones&Dangl,2006;J.Li et al., 2020).
In postharvest fruit and vegetable crops, these interactions become even more intricate due to the variable responses of the same host–ranging from immune to susceptible across different stages of development and ripening (Prusky &
Romanazzi, 2023), highlighting the complexity of host–pathogen dynamics.
Early in fruit development, host resistance prevents pathogen colonization and forces the microorganisms into quiescence, where noexternal symptoms are observed.
As the fruit ripens and senesces, host defenses weaken and susceptibility increases, allowing pathogen colonization to proceed (Cantu et al., 2008a,b).
These quiescent infections manifest in two distinct patterns:
- localized penetrating infection (LPI), where fungi breach specific fruit tissues at various developmental stages (Prusky, 1996); and
- non localized symptomless infections (NLSI), characterized by endophytic spread through vascular tissues, ultimately attacking the fruit at a distance from the initial infection site.
This manuscript focuses on well-characterized aspects of LPI, while the NLSI type of infection will not be covered in this review.
The LPI can be established in multiple locations of plant tissues, such as flower petals, receptacles, stamens, calyxes, fruit stems, and cuticles (Prusky, 1996).
These infections develop through either direct cuticle penetration or wound entry during host growth and handling until fruit maturation triggers symptom development.
Numerous fungal species, including Colletotrichum, Alternaria, Botrytis, Monilinia, Lasiodiplodia, a nd Phomopsis, have been documented to exist in this quiescent state within their hosts (Adaskaveg et al., 2000; Prins et al., 2000; Prusky et al., 2013).
Biotrophic pathogens such as Colletotrichum and Monilinia establish localized infections by penetrating the cuticle or taking advantage of minor wounds in cuticle and significant stem end wounds (Fig. 1).
These pathogens remain suppressed within the host cuticle or beneath the first layer of host cells until fruit ripening compromises the plant’s resistance.
Necrotrophs such as B. cinerea may penetrate flower stigmatic fluid before asymptomatic spread, whereas in kiwi fruit, it infects mainly through the stem end wounds (Adaskaveg et al., 2023).
Both biotrophic and necrotrophic pathogens show an early stage of quiescence during the unripe stage of fruit development (Prusky & Lichter, 2007; Petrasch
et al., 2019).
Several forms of quiescence have been identified:
- (1) delayed germination of conidia or postgermination growth arrest of
appressoria; - (2) asymptomatic endophytic growth in the apoplast; and
- (3) colonization of abscising floral organs, such as petals, followed by growth arrest in the ovaries or receptacles (Petrasch et al., 2019).
Ripening-induced susceptibility triggers pathogen activation, underscoring the importance of understanding quiescence mechanisms for effective postharvest disease control (Prusky, 1996; Ploetz, 2003; Galsurker et al., 2020).
Infection structures are key factors for either attaching and breaching the
intact cuticle or the wounded tissue for fungal infection.Spores that
germinated on the cuticle surface can form globular or lobate, melanized, or non melanized appressoria(Podilaetal.,1993; Lee& Bostock, 2006; Oliveira Lino et al., 2020).
In avocado, the long-chain fatty alcohols (≥ C24) present in the avocado
epicuticular wax critically stimulate Colletotrichum appressoria formation, whereas nonhost waxes suppress it (Podila et al., 1993; Prusky, 1996).
Upon germination, a needle-like hypha emerges from the base of the appressorium and punctures the cuticle.
After appressoria differentiation from germ tubes and penetration, the structure may either remain dormant within the cuticle– as observed in avocado (Daykin, 1984; Prusky, 1996)– or further develop into dendritic structures in the first cell layer beneath the cuticle, as seen in tomato (Alkan et al., 2015) (Fig. 1a).
Early infection typically avoids necrosis or visible host reactions, likely due to fungal effectors suppressing plant defenses (Kleemann et al., 2012; Giraldo & Valent, 2013); however, this may vary according to the host fruit.
Following its formation, the germinated appressoria in Colletotrichum, Monilinia, and Alternaria breach the cuticle through a tightly regulated process involving DNA replication, GTPase-mediated cell cycle progression (G1 to S
phase), and melanin deposition (Wao & K€oller, 1994; Fan & K€oller, 1998; Fetzner et al., 2014; Alkan et al., 2015; Tang et al., 2017;Liet al., 2022).
In B. cinerea, quiescent infections can occur without appressoria or with either unicellular or multicellular infection cushions (ICs).
These non melanized structures penetrate host tissues within 24–48 h of spore germination across various plant organs (Adaskaveg et al., 2000; Haile et al., 2019, 2020; Petrasch et al., 2019; Choquer et al., 2021;Biet al., 2023).
Most Botrytis infections in strawberries begin when spores germinate on flower
organs (primary infections) during or right after flowering, allowing hyphae to grow into the receptacle (Bristow et al., 1986). Infected senescent petals, stamens, and calyxes can facilitate primary infections in fruit (Petrasch et al., 2019).
In other cases, the stigmatic fluid provides nutrients for airborne conidia,
facilitating their growth in the humid microclimate in the raspberry (Williamson et al., 2007). The infection then spreads to the receptacle before becoming arrested and entering a quiescent state (Bristowetal., 1986; Jarvis, 1994; Prusky, 1996).
In grapes, a direct germination and infection of Botrytis was linked to cuticle
thickness, which restricts water and nutrient diffusion through water permeability sites (Herzog et al., 2015). While cultivars with thinner hydrophobic wax layers promote surface water and nutrient spread, increasing infection rates.
In other described cases of Botrytis, spores may form appressoria-like structures, from which hyphae can emerge at nearly perpendicular angles (Fig.2a) (Bietal., 2023).
Penetration is driven by turgor pressure, supported by the actin cytoskeleton, and is accompanied by the secretion of effectors, including phytotoxins,
proteases, and cell wall–degrading enzymes.
Unlike melanized appressoria in other fungi, B. cinerea depends more on enzymatic degradation than mechanical force for penetration.
Early infection triggers grapevine defenses, including antimicrobial proteins and
cell wall reinforcement, which suppress fungal growth until favorable conditions reactivate pathogenesis (Haile et al., 2019).
In summary, postharvest pathogens first detect the host and then regulate diverse metabolic processes to:
- develop penetration structures;
- identify optimal entry sites;
- equip these structures with cutinase and lipases to breach the host cuticle, ensuring successful quiescence; and
- inhibit fungal colonization and host immunity until fruit ripening.
Therefore, understanding this host–pathogen mechanism during quiescence and its activation proves critical for the prevention of fruit decay throughout
postharvest storage life (Bruton et al., 1998; Prusky et al., 2010).
Contents
The dynamics of the quiescent infection
Quiescence in Colletotrichum
Quiescence in Botrytis
Quiescence in Monilinia
Quiescence in pathogens that penetrate through wounds
Quiescence exit and virulence onset
Conclusion
Postharvest pathogens infect produce at pre- and postharvest conditions, entering quiescence until ripening weakens host defenses.
Pathogens employ melanized appressoria, turgor pressure, and nutrient-sensing to breach cuticles.
Biotrophs persist beneath the cuticle of host cells, whereas necrotrophs like B. cinerea employ cell death–inducing factors, enhanced by nutrient-rich target sites, to facilitate penetration but remain dormant until ripening, activating only under favorablec onditions.
The questionis whether quiescence is a fungal inhibition process or an immune response of the unripe host fruit.
Despite factors that inhibit fungal penetration and influence quiescence conditions, biotrophic pathogens such as Colletotrichum sense their environment and reprogram their activity through chromatin regulation.
By contrast, necrotrophs, such as Botrytis and Penicillium, maintain quiescence in locations tied to nutrient availability, which enables the expression of early key virulence factors that may regulate quiescence activation stages.
Host responses during quiescence include genes for cell recognition, and ET signaling– usually active in green fruit– alongside defensegenes (e.g.PR-10), with JA/ET synergizing defenses and ABA inducing epidermal callose deposition, and upregulated antifungal compounds that all together prevent the activation of quiescence.
Colletotrichum’s biotrophic hyphae secrete effectors (e.g. ChELP2) interacting with host PRRs, probably contributingto the modulation of quiescence.
In necrotrophic pathogens, NPR1, a central regulator of plant defense, acts as an SA receptor and fine-tunes JA- and ET mediated resistance against necrotrophs by upregulating defense related genes as PR proteins and b-1,3-glucanases.
Across different pathotypes, the onset and progression of fruit ripening serve as the primary trigger for pathogens to shift from host immune recognition to an active necrotrophic lifestyle.
However, the loss of quiescence in ripening fruit appears to result not from distinct response mechanisms in susceptible fruits but from a diminished expression of the same defense-related pathways.
Emergence from quiescence is attributed to:
- greater access to cell wallsubstratesfromfruitsoftening;
- weakenedhostdefenses;
- reduced antifungal compounds; and
- activation of virulence factors modifying the host. Inhibition of fungal growth during the fruit’s immune response remains strongly dependent on host metabolism.
Inhibition of fungal growth during the fruit’s immune response remains strongly dependent on host metabolism.
A deeper understanding of the mechanisms underlying fruit immunity during quiescence is therefore critical to preventing pathogen resurgence, which is a major cause of postharvest diseases and losses reaching up to 60% of harvested crops.
Picture is Fig. 1 from the original paper - Colletotrichum and Monilinia establish quiescent infections in fruit either through direct penetration or minor wounds, using hyphae originating from germinating spores.
Colored appressoria indicate melanization of the structure.
Darker staining of host cells signifies cells in the process of dying.
Germinated hyphae remain dormant beneath the cuticle or penetrate one to two cell layers, penetrating to cells using their biotrophic capability and further forming dendritic structures before the cells become necrotic.
The limited number of dead cells at each infection site prevents the formation of visible necrotic lesions during quiescent infection.
Wound penetration occurs without appressorium formation.
The accumulation of reactive oxygen species (ROS) in wounded tissue causes molecular damage, ultimately leading to cell death and facilitating initial fungal colonization (Wang et al., 2019).
Following this process, the direct biotrophic penetration of cells occurs. Colletotrichum will secrete novel cell death-inducing effector proteins (Takahara
et al., 2021; Wang et al., 2024), but they are mainly produced when the fungus is released from quiescence.
Source
Quiescence of postharvest pathogens: a fungal inhibition process or an
immune response of the unripe host fruit?
Dov B. Prusky, Tong Chen, Shiping Tian, Yong Chen and Boqiang Li
NewPhytologist (2026)
doi: 10.1111/nph.70913













