News
How does blue mold affect citrus fruits?
The research by E. Silva et al. analyzes the virulence mechanisms of Penicillium italicum, the blue mold of citrus fruits

Pathogenic fungi cause significant post-harvest losses in fruit production chains, reducing commercial value and threatening food security.
Citrus fruits, which are highly valued and widely consumed, are particularly affected. Transportation, distribution, and long storage periods make these fruits highly susceptible to microbial decay.
Penicillium, principales patógenos en la poscosecha de los cítricos
Dos especies de Penicillium, P. digitatum y P. italicum, son los principales agentes causantes del moho verde y azul, respectivamente, y juntas son responsables de la gran mayoría del deterioro poscosecha de los cítricos.
Mientras que P. digitatum se considera la especie más agresiva y prevalente, responsable de hasta el 90 % de las pérdidas, P. italicum plantea un reto particular debido a su tolerancia a las bajas temperaturas y a la actividad del agua. Esta resistencia fisiológica le permite persistir en condiciones de almacenamiento menos favorables que P. digitatum, lo que facilita su rápida propagación y contribuye a pérdidas significativas.
La gestión actual de las enfermedades poscosecha de los cítricos se basa en gran medida en la aplicación de fungicidas sintéticos.
Sin embargo, la aparición cada vez mayor de cepas de Penicillium resistentes a estos productos, junto con la creciente presión regulatoria y pública para encontrar alternativas más seguras y sostenibles, ha creado una necesidad urgente de nuevas estrategias de control.
Understanding the mode of action to improve control strategies
To this end, understanding the molecular basis of fungal infection is crucial for identifying new strategies to mitigate postharvest diseases.
In recent years, research has been conducted into the molecular mechanisms underlying the virulence of P. digitatum, including the identification of specific effectors, transporters, and secondary metabolites. However, the biology of P. italicum remains less well understood.
Delving deeper into the biology of P. italicum
In phytopathogenic fungi, virulence is multifactorial and often involves a combination of enzymatic tissue degradation, resistance to environmental stress, and weakening of host defenses.
Secondary metabolites, in particular, are recognized as critical virulence factors in many plant-pathogen interactions. These low molecular weight bioactive compounds, synthesized by biosynthetic gene clusters, can serve as phytotoxins, plant immunity suppressors, antimicrobial agents, or signaling molecules involved in host colonization.
Biosynthetic gene clusters often encode enzymes such as nonribosomal peptide synthetases and polyketide synthetases, which produce structurally diverse and biologically active metabolites.
Secondary metabolites of P. italicum
Although the contribution of secondary metabolites of P. italicum to virulence has not been fully characterized, recent studies have revealed that this species produces a distinctive repertoire of bioactive compounds.
For example, P. italicum has been reported to synthesize piperazinoazepines, the pyranone 2,5-dihydro-4-methoxy-2H-pyran-2-one, and italic acid, a tetrahydrofuran derivative.
In addition, researchers have performed in vivo metabolomic analyses of oranges infected with P. italicum and identified the presence of diketopiperazine alkaloids, including brevianamide F, which contribute to microbial antagonism against endophytes.
Functional genetic approaches, such as gene inactivation or silencing, provide effective tools for analyzing the role of secondary metabolites in fungal virulence.
Importance of direct gene/function analysis
Although secondary metabolites are often assumed to contribute to pathogenicity, their effects can be highly context-dependent. For example, deletion of the tqaA gene in P. digitatum, responsible for the biosynthesis of tryptochylanine A, does not affect the fungus's ability to infect citrus fruits, despite the toxicity of the compound.
This underscores the importance of directly analyzing gene-function relationships, rather than relying solely on metabolite abundance or gene identification.
Research into the functional role of cyclic peptides and discovery of a potential molecular target
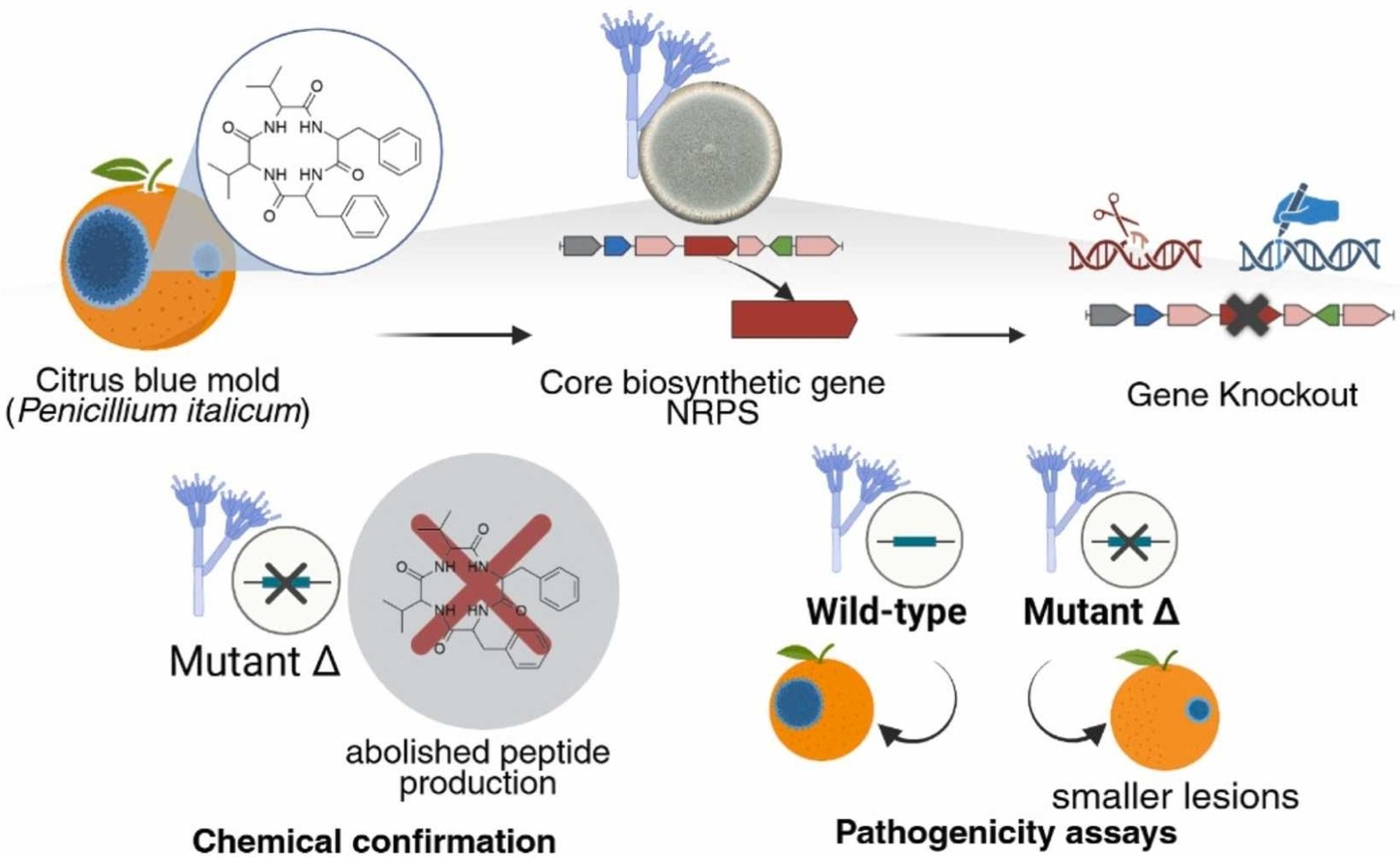
A recent study investigated the functional role of cyclic peptide secondary metabolites in the infection process of P. italicum in citrus fruits.
Through LC-HRMS (Liquid Chromatography-High Resolution Mass Spectrometry) analysis of infected fruit tissue, a unique group of cyclic peptides was identified, including fungisporin and structurally related tetrapeptides produced during host colonization.
Using a targeted gene deletion strategy, the hcpA gene responsible for the biosynthesis of nonribosomal peptide synthase (NRPS) enzymes was altered. This eliminated the production of these metabolites, confirming their essential role in biosynthesis.
Although the ΔhcpA mutant showed normal development and sporulation, it was more susceptible to osmotic stress and caused smaller lesions on oranges, indicating reduced virulence.
This study identifies a promising molecular target for the development of innovative approaches to the control of post-harvest citrus diseases and reveals a direct connection between secondary metabolism and the infection capacity of P. italicum, the causal agent of blue mold.
Graphic summary according to Silva, E. et al, 2026
Sources
Silva, E.; Barbosa, J. C.; Gárcia, A. G.; Berlinck, R. G.; S.; Ballester, A. R.; Candelas, L. G.; Fill, T. (2026).
Decoding virulence in Penicillium italicum: A functional link between NRPS-derived cyclic peptides and citrus infection
Postharvest Biology and Technology, 234: 114069.
https://www.sciencedirect.com/science/article/abs/pii/S0925521425006817 Access on 24/01/2026.
https://doi.org/10.1016/j.postharvbio.2025.114069 Access on 24/01/2026.
Cover Photo
Pacific Pests, Pathogens & Weeds, https://apps.lucidcentral.org/pppw_v10/text/web_full/entities/citrus_storage_moulds_197.htm, access on 25 enero 2026














